外周神经损伤(Peripheral Nerve Injury, PNI)是临床中常见且致残率较高的神经系统疾病,广泛存在于创伤、肿瘤切除及医源性损伤等场景中。尽管显微外科缝合、神经导管移植及电刺激治疗等手段已被应用于临床,但由于神经损伤区域空间受限、个体差异显著以及术后炎症反应复杂,现有治疗方式在精准调控、长期稳定性和微创性方面仍面临诸多挑战。尤其是传统神经接口多依赖刚性器件或有线连接,难以与柔软、动态变化的神经组织实现长期共形贴附,限制了其在慢性神经修复中的应用。随着柔性电子与生物可吸收材料的发展,能够在体内实现无线操控、形态自适应并在完成治疗后自然降解的新型神经接口,成为外周神经精准治疗领域的重要发展方向。然而,如何在保证器件力学顺应性的同时,实现对局灶性神经区域的稳定包覆及多模态治疗功能,仍是亟待解决的关键科学与工程问题。
针对上述挑战,复旦大学智能机器人与先进制造创新学院/智慧纳米机器人与纳米系统国际研究院的梅永丰/宋恩名研究团队报道了一种自卷绕、生物可吸收的双稳态神经接口(self-wrapping bistable, SWB neural interface),用于局灶性外周神经损伤的无线多模态治疗。该器件基于应力梯度驱动的超薄 SiNx 双层结构,在触发后可由平面状态自发转变为三维卷绕结构,从而对不同直径的外周神经实现温和而稳定的包覆。在功能集成方面,该神经接口引入MXene 光热层与药物负载模块,构建了无线近红外触发的光热治疗与药物释放相结合的多模态治疗体系,实现了对神经修复过程的时空精准调控。通过系统的力学建模、有限元仿真及在体实验,研究团队验证了器件自卷绕双稳态转变的可靠性、生物可吸收特性及其在大鼠坐骨神经损伤模型中的治疗效果。相关成果以 “A self-wrapping, bioresorbable neural interface for wireless multimodal therapy of localized peripheral nerve injury” 为题发表于 Proceedings of the National Academy of Sciences of the United States of America(PNAS)。
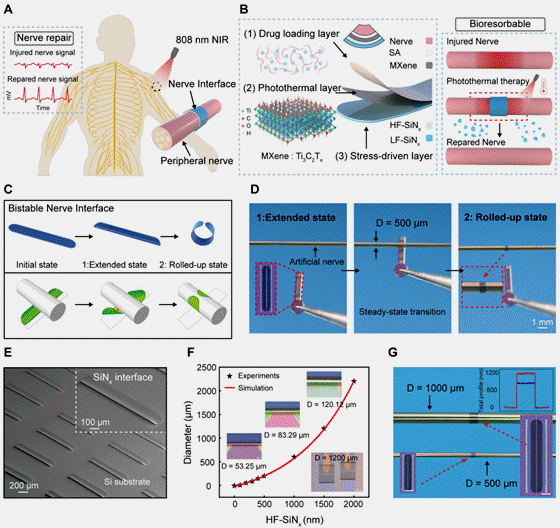
图1:自卷绕双稳态神经界面的结构设计与工作原理
该神经界面由具有内应力梯度的SiNx 双层薄膜构成,在释放约束后可由平面结构自发卷绕形成三维管状结构。器件在展开与卷绕状态之间呈现明显的双稳态特征,使其在植入过程中保持平面、在目标区域实现自适应包覆。该设计有效避免了传统缝合或外力固定方式对神经组织造成的二次损伤。
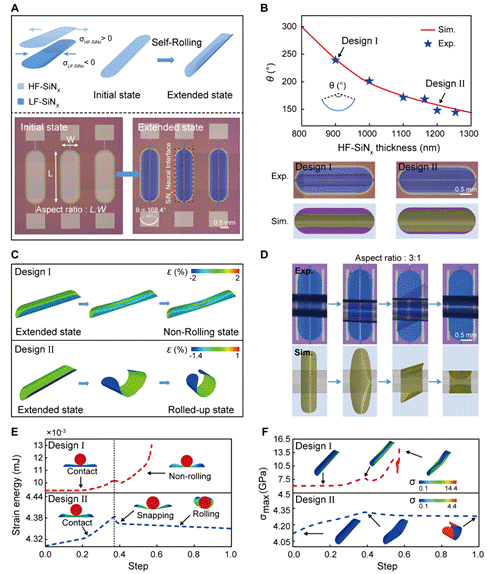
图2:自卷绕过程的力学机制与可调控性分析
复旦大学智慧纳米机器人与纳米系统国际研究院刘鹏川博士为该论文第一作者,该工作得到了国家自然科学基金等项目的资助和支持。
文章信息:
P. C. Liu#, L. J. Zhou#, D. Xu#, D. Q. An, Y. F. Lu, B. F. Hu, Y. T. Shao, N. G. Huang, C. J. Guo, L. Chen, J. B. Li, J. H. Li, F. Y. Liang, J. H. Liu, G. S. Huang, Y. F. Mei, R. Li*, E. M. Song*. A self-wrapping, bioresorbable neural interface for wireless multimodal therapy of localized peripheral nerve injury. Proc. Natl. Acad. Sci. U. S.A., 123(2): e2521817123(2026).
原文链接:https://www.pnas.org/doi/10.1073/pnas.2521817123
A self-wrapping, bioresorbable neural interface for wireless multimodal therapy of localized peripheral nerve injury
High-precision in vivo therapeutic technologies that establish three-dimensional (3D), multimodal neural interfaces with targeted biotissues offer significant clinical potential for the timely treatments of localized peripheral nerve injury (PNI). Current approaches for this purpose such as implantable devices face challenges in terms of percutaneous wires and/or nondegradable designs, and support only single-mode operation that lack microscale spatial resolution. Here, we develop a miniaturized, self-wrapping system that yields wireless, multimodal neural interfaces with 3D adaptation across localized peripheral nerves at scales ranging from tens of micrometers (15 μm) to millimeters. Such platform integrates multilayer architectures that include SiNx layers as the mechanically triggered substrate for 3D wrapping, with multimodal treatments via MXene and drug-loaded layers for photothermal stimulation and pharmacological release. Experimental and computational studies establish operational principle as the basis for the combination of long-term photothermal therapy and transient drug delivery at high spatiotemporal resolution. In vivo tests on living rat models demonstrate that the implantable neural interface can roll up across the localized, dynamic surface of injured nerves, providing sustained treatments over 1 mo in a fully bioresorbable design after the healing process. These findings create future opportunities of such wireless, multimodal system with 3D self-wrapping techniques for precise PNI therapeutic strategies.
